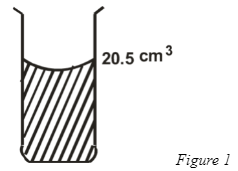
- The figure 1 below shows a measuring cylinder with a fluid to the level shown.
Find the new level when a solid of mass 13.6g and a density 1250kgm−3 is fully immersed into the cylinder.
(2 marks) - Define current and state its SI units. (2 marks)
- In a hydraulic press, a force of 200N is applied to a master piston of area 25cm3. If the press is designed to produce a force of 5000N, determine; The area of the slave piston (2mks)
- A steel needle when placed carefully on water can float. When detergent is added to the water it sinks. Explain (2mks)
- Water is known to boil at 100°C. A student heated some water and noticed that it boiled at 101°C. State two possible reasons for this observation (2mks)
- Explain the cause of random motion of smoke particles as observed in Brown Motion experiment using a smoke cell (3mks)
- Name two types of forces that act between bodies not in contact. (2marks)
- Convert the each of the following temperatures into Kelvin . (2 marks)
- 25°c
- -123°c
- Calculate the maximum pressure a glass block of density 2.5gcm-3 would exert on a horizontal surface, if the block measures 20cm x 10cm x 5cm. (3marks)
- Give a reason why fish can survive under water in a pond even when the surface is frozen. (1mark)
- Give one advantage of alcohol over mercury as a thermometric liquid. (1mark)
- An object weighs 49N on earth where acceleration due to gravity is 9.8N/kg. Find the acceleration due to gravity on another planet where the same object weighs 40.5N. (2 marks)
- Give a reason why boiling water cannot be used to sterilize a clinical thermometer.(1 mark)
- State four factors that affect thermal conductivity. (4 mks)
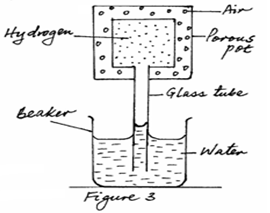
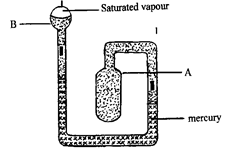
- Figure 3 below shows apparatus used to compare the rates of diffusion of gases.
Water is observed to rise up the glass tube. Explain this observation.(2 marks) - The mercury column in a barometer is 760mm high. Taking the density of mercury to be 13.6g/cm³, calculate the atmospheric pressure in N/m².(2 marks)
-
- What is surface tension? (1 mark)
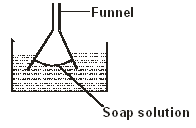
- Figure 4 shows a funnel dipped into a liquid soap solution.
Explain what happens to the soap bubble when the funnel is removed from the soap solution. (2 marks)
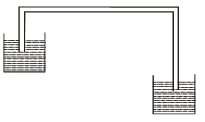
- Figure 7 shows a liquid being siphoned from a beaker to another.
Figure 7- Indicate on the diagram the direction of flow of the liquid. (1 mark)
- Explain what would happen to the flow of the system if it was put in a vacuum.(2 marks)
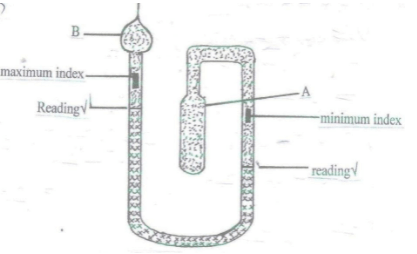
- The diagram below shows a six’s maximum/ minimum thermometer.
- What is the thermometric liquid in this thermometer? (1mark)
- Explain how the thermometer indicates maximum and minimum temperature. (4 marks)
- Indicate on the diagram the two points where the reading of the temperature shown by the thermometer can be made
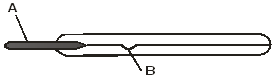
- The figure below shows a clinical thermometer which is not graduated.
- Name the parts indicated with letters A and B. (2 marks)
- Give the range of the scale for the above thermometer. (1 mark)
- A mercury barometer reads 760mmHg at sea level and 700mmHg at the top of a mountain. If the density of mercury is 13,600kg/m3 and average density of air is 1.30kg/m3, calculate the height of the mountain.(3 mks)
- In a vacuum flask, the walls enclosing the vacuum are silvered on the inside. State the reason for this.(1mk)
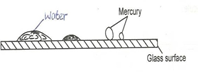
- The figure below shows the shapes formed when drops of water and mercury are placed on the surface of a clean glass plate.
Explain the difference in the shapes. (2mks) - Explain why air is not used as a brake fluid. (1mk)
- 20cm3 of water is mixed with 30cm3 0f liquid L. Calculate the density of the mixture given that the density of water is 1000kg/m3 and that of liquid L is 800kg/m3. (3 mks)
- State four uses of the Electroscope (4mks)
- An object of height 5m is placed 10m away from a pinhole camera. Calculate:
- The size of the image if its magnification is 0.01. (2 mrks)
- The length of the pinhole camera (2 mks)
- What do you understand by the term temperature ( 1 mk)

MARKING SCHEME
- V = m = 13.6 = 10.88cm3. ✔1
p 1.25
New reading = 20.5 + 10.88 = 31.38cm3 ✔1 - Is the flow of charge (charge per unit time) SI units Ampere “A”
- F₁ = F₂
A₁ A₂
200N = 5000N
25cm2 A₂
A2 = 5000N
200
= 625cm2 - The surface of the water behaves like a thin stretched elastic skin. So the needle will float. When detergent is added, it breaks the surface tension of the water making it to sink
-
- Atmospheric pressure is higher than normal
- Presence of impurities/addition of impurities
- They move in continuous random movement because of uneven bombardment by the invisible particles or molecules of the air.
-
- Magnetic force
- Electrostatic
-
- T=X + 273
=25 + 273
=298k - T= −123 +2 73 =150K
- T=X + 273
- Mass = 20 x 10x 5 x 2.5
= 2500g = 2.5kg
P = F
A
= 25 x 104
50
= 5000Nm-2 - Anomolous expansion of water
- Alcohol can measure lower temperatures than mercury
- Mass of the object = 49 = 5kg ✔
9.8
Acceleration due to gravity 40.5 = 8.1N/kg (2mks)
5 - Boiling water would burst a clinical thermometer. ✔ (1mk)
-
- Temperature difference between the ends of the conductor
- The length of the conductor
- Cross-section area of the conductor
- Nature of the material
- Hydrogen diffuses out of the porous pot faster than air diffuses into the porous pot.✔
The pressure inside the porous pot reduces and the higher atmospheric pressure pushes the water up the glass tube. ✔ (2mks) - Atmospheric pressure = 13,600 x 0.76 x 10 ✔
= 103360N/m² ✔ (2mks) -
- It’s the force that makes a free liquid surface behave like a stretched elastic skin ✔1
- Bubble flattens to a film1 and move up in the funnel in order to make its surface area as small as possible due to surface tension ✔1
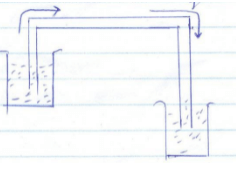
- The flow stops because there is no atmospheric pressure to provide pressure difference ✔1
-
- Alcohol ✔
-
- When the temperature rises, bulb A expands pushing the mercury down on that side which in turn pushes the index up on the other side so that maximum temperature is recorded ✔
- When temperature goes down the vapor pressure in bulb B pushed the alcohol above the mercury down which in turn pushes the mercury down. The index is left at the maximum point it had reached. The index on the other side is now pushed up and will be left at the highest point (minimum) when the situation reverses ✔
-
-
-
- A – Bulb
- B – Constriction
- 35°C – 43°C
-
- Difference in pressure 760 – 700 = 60mm✔1
60/1000 x 13600 x 10 = h x 1.3 x 10 ✔1
h = 8160
1.3 ×10
h = 627.69 m✔1 - To prevent heat loss/ gain through radiation. ✔1mk
- For mercury cohesion force is greater/stronger than adhesion forces hence the drops are spherical for water the adhesion forces are greater than /stronger than cohesion forces hence water on the grass surface spreads wetting it.
- Air will be compressed
- 20cm3 of water is mixed with 30cm3 0f liquid L. Calculate the density of the mixture given that the density of water is 1000kg/m3 and that of liquid L is 800kg/m3. (3 mks)
Volume of the mixture = 20 + 30 = 50cm3
Mass of water = 20 x 1 = 20g
Mass of L = 0.8 x 30 =24g
Total mass = 44g;
Density = mass
volume
Density = 44/50
= 0.88 g/cm3 -
- To detect the presence of charge on a body
- To test the sign of the charge on a charged body
- To test the quantity of charge on a charged body
- To test for insulation properties of a material
-
- Magnification = height of image/height of object
0.01 = hi/5
Hi = 0.01×5
=0.05 m
The image is 5 cm high - magnification = image distance (v)
object distance (u)\
0.01 = v/10
V = 0.01 × 10
= 0.1 m
The length of the pinhole camera is 10 cm
- Magnification = height of image/height of object
- It’s the degree of coldness or hotness of a body on some chosen scale
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Physics Questions and Answers - Form 1 End Term 3 2022 Exams.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students