The table gives standard reduction potentials for some half cells.
| Half cell | Half cell equation | E0/V |
| I | Fe3+(aq) + e → Fe2+(aq) | +0.77 |
| II | K+(aq) +e → K(s) | −2.92 |
| III | Ag+(aq) + e → Ag(s) | +0.80 |
| IV | Pb2+(aq) +2e → Pb(s) | −0.13 |
| V | l2(aq) + 2e → 2l−(aq) | +0.54 |
- State the standard conditions of an electrochemical cell.
- An electrochemical cell was constructed using half-cells III and IV.
- Complete Figure 2 by labelling the parts of the cells indicated as A1- A4
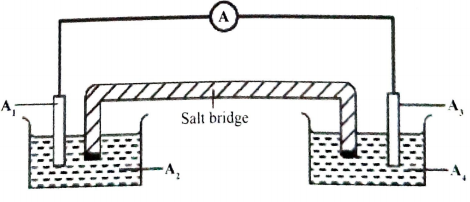
Figure 2 - Write an equation for the cell reaction and calculate the e.m.f. of the cell.
- The salt bridge helps in completing the circuit. Explain why a saturated solution of potassium chloride is not suitable for use in the salt bridge in this electrochemical cell.
- State why it is not possible to construct a similar electrochemical cell using half-cells II and III.
- State and explain the observations made when aqueous potassium iodide is added to aqueous iron(III) sulphate.
- Acidified potassium dichromate(VI) and acidified potassium manganate(VII) may be used in determining concentration of Fe2+ ions in a sample. If acidified potassium dichromate(VI) is used, an indicator is added to determine the end point but for acidified potassium manganate(VII), no indicator is added.
- Explain why it is not necessary to use an indicator when acidified potassium manganate(VII) is used.
- An alloy containing iron was dissolved in an acid and the total volume made up to 250cm3. 25.0 cm3 of this solution required 18.0 cm3 of 0.15 M acidified potassium dichromate(VI) to react completely. The equation for the reaction is:
Cr2O72− (aq) + 6Fe2+(aq)+ 14H+(aq) →2Cr3+(aq)+ 6Fe3+(aq) +7H2O(l)
Calculate the mass of iron in the alloy (Fe = 56.0).