The Figure below shows an energy level diagram for the decomposition of hydrogen peroxide using a catalyst
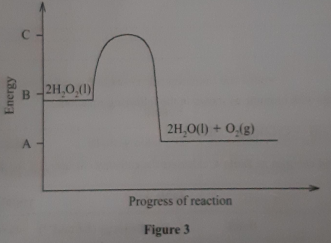
- Using the energy values A, B and C, write an expression for
- ΔH of the reaction
- activation energy.
- On the same axis., sketch a curve that would be obtained if the reaction was carried out without a catalyst.