Questions
- The table below shows solutions A, B and C are tested and observations records as shown:
Solution Observations on indicator A Methyl orange turns yellow B Phenolphthalein turns colourless C Litmus turns purple - Using the table above, name an acid
- How does the pH value of 1M potassium hydroxide solution compare with that of 1M aqueous ammonia? Explain
- The information below gives PH values of solutions V, W, X, Y, Z
Solution PH values V
W
X
Y
Z2
6.5
11
14
4.5- Which solution is likely to be:
- Calcium hydroxide?
- Rain water?
- Which solution would react most vigorously with Zinc carbonate
- Which solution is likely to be:
-
- Complete the table below to show the colour of the given indicator in acidic and basic solutions.
Indicator Colour in Methyl Orange Acidic Solution Basic Solution Phenolphthalein Colourless - How does the PH value of 0.1M potassium hydroxide solution compare with that of 0.1M aqueous ammonia? Explain.
- Complete the table below to show the colour of the given indicator in acidic and basic solutions.
- Use the information given below to answer the questions that follow:
Solution G H I J K pH 1.5 6.5 13.0 7.0 8.0 - Which of the solutions would be used to relieve a stomach upset caused by indigestion?
- Which solution is likely to be:
- Dilute sulphuric acid?
- Sodium hydroxide solution?
- Solid copper (II) oxide is a base although it does not turn litmus paper to blue. Explain
- Below are the pH values of 4 types of medicine represented by letters P, Q, R and S
MEDICINE pH VALUES P
Q
R
S7.0
5.0
8.0
6.0- It is not advisable to use S when a patient has indigestion .Explain
- What is the role of chemistry in drug manufacture
- Explain why very little Carbon (IV) oxide gas is evolved when dilute sulphuric (VI) acid is added to lead (II) carbonate
- State one commercial use of Calcium Oxide
- The following data gives the pH values of some solutions
Solution pH P
Q
R14.0
6.8
2.5- What colour change would occur in solution P on addition of two drops of phenolphthalein indicator?
- State the pH value of a resulting solution when equal moles of solution P and R react
- In an experiment, ammonia gas was prepared by heating ammonium salt with an alkali.
After drying, ammonia gas was collected at room temperature and pressure.- What is meant by the term alkali?
- Explain using physical properties of the gas why ammonia is not collected by downward delivery
- The table shows the colours obtained when some indicators are added to solutions:-
Solution Blue litmus paper Indicator W Distilled water ………………….. Colourless Calcium hydroxide Blue Pink Nitric acid ………………………… Colourless - Complete the table by filling in the missing colours
- Identify indicator W
- Flower extracts can be used as Acid-base indicators. Give two limitations of such indicators
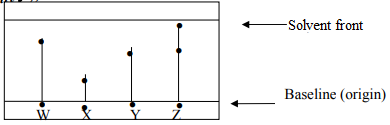
- The diagram below shows spots of pure substances W, X, and Y on a chromatography paper. Spot Z is that of a mixture
After development W, X, and Y were found to have moved 9cm3, 4cm3 and 7cm3 respectively. Z has separated into two spots which have moved 7cm3 and 9cm3:-
On the diagram:-- Label the baseline and solvent front
- Show the position of all the spots after development
- Identify the substances present in mixture Z
- A beekeeper found that when stung by a bee, application of a little solution of sodium hydrogen carbonate helped to relieve the irritation of the affected area. Explain
- 10g of sodium hydrogen carbonate were dissolved in 20cm3 of water in a boiling tube. Lemon juice was then added dropwise with shaking until there was no further change.
- Explain the observation which was made in the boiling tube when the reaction was in progress
- What observations would be made if the lemon juice had been added to copper turnings in a boiling tube?
- Complete the table below to show the colour of the given indicator in acidic and basic solutions:
Indicator Colour in acidic solution Basic Solution Methyl orange Pink Phenolphthalein Pink - Solutions can be classified as acids, bases or neutral. The table below shows solutions and their pH values:-
Solutions pH Values K 1.5 L 7.0 M 14.0 - Select any pair that would react to form a solution of PH 7
- Identify two solutions that would react with aluminium hydroxide. Explain
Answers
-
- B
- pH of potassium hydroxide is higher than that of aqueous ammonia. KOH ions are dissociated more than that of aqueous NH3
-
-
- X
- W
- V
-
-
- Methyl Orange Red/Pink ✓½
Phenolphthalein Colourless/Pink ✓½ - The PH of 0.1M KOH is higher than of 0.1M aqueous ammonia ✓1
KOH is strongly dissociated in solution. ✓1
- Methyl Orange Red/Pink ✓½
-
- K
-
- G
- I
- Copper (II) oxide is insoluble in water hence there are no OH- ions in the mixture
-
- S is acidic and would make the situation worse ✓ ½
- Discovery of drugs processing and testing is the work of chemists. Chemists are professionals who have studied chemistry ✓ ½
- Its due to formation of insoluble Lead(II) carbonate hence preventing any further reaction.
- CaO is used in correcting soil acidity. √ 1
-
- Pink
- 7.0
-
- alkali is soluble base. √ 1
- Because it is lighter than air. √ 1
-
-
Solution Blue litmus paper Indicator W Blue Red - Phenolphthalein
-
-
- -give inconsistent results√ ½
-expire shortly√ ½ -
- Maximum sports-award 1 ½ mks
Fail any one- award - W √ ½ and Y √ ½
- -give inconsistent results√ ½
- Sting of a bee is acidic √ 1 and is neutralized by sodium hydrogen carbonate√ ½ into a salt, carbon IV) oxide and water. This gives pain relief. √ ½
-
- There was production of effervescence . The lemon juice contain an acid that reacts with the carbonate to produce carbon (IV) oxide.
- No production of bubbles. Copper is below hydrogen in the reactivity
-
- Yellow
- Colourless
-
- K and M
- K and M
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Acids, Bases and Indicators Questions and Answers - Chemistry Form 1 Topical Revision.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students