- You are provided with:
Acid A labeled solution A;
2.0 M sodium hydroxide solution labeled solution B.
Solution C containing 25.0 g per litre of alkanoic acid.
You are required to:- Prepare a dilute solution of sodium hydroxide, solution B
- Determine the:
- Molar mass of the alkanoic acid.
- Reaction ratio between sodium hydroxide and acid A.
Procedure 1
Using a pipette and a pipette filler, place 25.0cm3 of solution B in a 250.0 ml volumetric flask. Add about 200 cm3 of distilled water. Shake well. Add more distilled water to make up to the mark. Label this solution D. Retain the remaining solution B for use in procedure II.
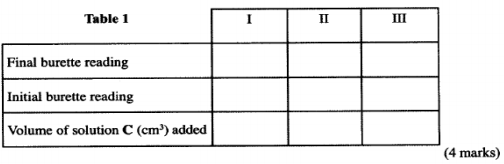
Fill a burette with solution C. Using a clean pipette and a pipette filler, place 25.0 cm3 of solution D into a 250 ml conical flask. Add two drops of phenolphthalein indicator and titrate with solution C. Record your results in table 1. Repeat the titration two more times and complete the table.
Determine the:
(i) Average volume of solution C used; (1 mark)
(ii) Concentration of solution D in moles per litre; (1 mark)
(iii) Concentration of the alkanoic acid in solution C in moles per litre (1 mole of the acid reacts with 3 moles of the base); (1 mark)
(iv) Molar mass of the alkanoic acid. (1 mark)
Procedure II
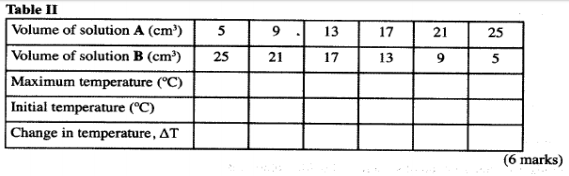
Fill a clean burette with solution A. Place 5 cm3 of solution A into a 100 ml beaker. Measure the initial temperature of solution A in the beaker and then record it in table II. Using 10 ml or 100 ml measuring cylinder, measuring 25 cm3 of solution B. Add it to solution A in the beaker and immediately stir the mixture with a thermometer. Record the maximum temperature reached in table II. Repeat the experiment with other sets of volumes of solution A and B and complete the table.
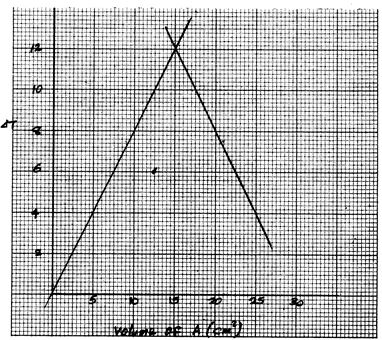
(a) On the grid provided, plot graph of ΔT (Vertical axis) against the volume of solution A. (3 marks)
(b) From the graph, determine the volume of solution A which gave the maximum change in temperature. (1 mark)
(c) Determine the volume of solution B that reacted with the volume of solution A in (b) above. (1 mark)
(d) Calculate the:
(i) Ratio between the volumes of solutions A and B that neutralised one another. (1 mark)
(ii) Concentration in moles per litre of the acid in solution A.
(Assume that the volume ratio is the same as the mole ratio). (1 mark)
- You are provided with solids E, F and G.
Carry out the tests below and write your observations and inferences in the spaces provided.- Place all of solid E in a boiling tube. Add 20 cm3 of distilled water and shake until all the solid dissolves. Label this as solution E.
- To about 2 cm3 of solution E in a test tube, add four drops of 2 M sulphuric (VI) acid.
- To about 2 cm3 of solution E in a at test-tube, add 2 M sodium hydroxide dropwise until in excess.
- Place one half of solid F in a test tube. Add 2 cm3 of distilled water and shake well. Add 4 drops of this solution to about 2 cm3 of solution E in a test-tube.
- To about 2 cm3 of solution E in a test tube, add two drops of aqueous potassium iodide.
- To about 2 cm3 of solution E in a test tube, add four drops of 2 M sulphuric (VI) acid.
- Using a metallic spatula, ignite about one half of solid G in a Bunsen burner flame.
- Place the other half of solid G into a boiling tube.
Add 15 cm3 of distilled water and shake well. Label this solution G.
Use this solution for the following tests.- Place 2 cm3 of solution G in a test tube and determine its pH.
- To about 2 cm3 of the solution obtained in (ii) above, add 3 drops of acidified potassium manganate (VII).
- To about 2 cm3 of the solution obtained in (ii) above, add 2 drops of bromine water.
- Place 2 cm3 of solution G in a test tube and determine its pH.
- To the remaining solution G in the boiling tube, add the other half of solution F.
- Using a metallic spatula, ignite about one half of solid G in a Bunsen burner flame.
- Place all of solid E in a boiling tube. Add 20 cm3 of distilled water and shake until all the solid dissolves. Label this as solution E.
Marking Scheme
-
I II III Final burette reading 13.80 27.80 40.70 Initial burette reading 0.00 13.80 27.30 Volume of solution used (cm3) 13.80 13.50 13.40
Average volume used
13.50 + 13.40 = 13.45 cm3
2
Mava = Mbvb
2 x 25 = 250 x Vb = 0.20M
Moles of NaOH used = 0.2 x 25/1000 = 0.005 moles
Moles of acid used = 1/3 x 0.0005
Concentration of acid = 0.005 x 100 = 0.12M
13.45 x 3
Molar mass of acid = 25/0.12 = 208.3
Table 2
Volume of solutin A (cm3) 5 9 13 17 21 25 Volume of soluton B (cm3) 25 21 17 13 9 5 Maximum temperature (°C) 30.5 34.0 36.5 36.5 34.0 30.5 Initial temperature (°C) 26.5 26.5 26.5 26.5 26.5 26.5 ΔT change in temperature 4.0 7.5 10.0 10.0 7.5 4.0 -
- 15cm3
- 30 - 15 = 15cm3
-
- 15:15 = 1:1
- MaVa = MbVb
Ma x 15 = 1
2 x 15 1
Ma = 2 x 15 = 2
15
Ma = 2M
-
-
a (i) Observations Inferences White ppt formed
No effervesenceCO2-3 and SO2-3 ion absent
Probably Pb2+, Ba2+ or Ca2+ may be present(ii) Observations Inferences White ppt which
Dissolves in excessPb2+ present (iii) Observations Inferences White ppt formed Insoluble cpd of Pb2+ is formed (iv) Observations Inferences Yellow ppt Pb2+ ions confrimed or Pbl2 formed b (i) Observations Inferences Burns with a smoky flame Unsaturated organic cpd or long chain hydrocarbon (ii) Observations Inferences Colourless solution, turns red pH 1- 2 Carboxylic acid present (iii) Observations Inferences Effrevesence colourless gas evolved
Odourless gasConfirm G was acid and F was carbonate I Observations Inferences Decolourised KMnO4(l) Unsaturated alkene or alcohol present II Observations Inferences Bromine water decolourised Unsaturated alkene present or alkyne
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download KCSE 2010 Chemistry Paper 3 Questions and Answers.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students