INSTRUCTIONS
- Answer all the questions in the spaces provided.
- Mathematical tables and silent non-programmable electronic calculators may be used
- All working must be clearly shown where necessary.
- Candidates should answer the questions in English.
For examiners use only.
|
Question. |
Maximum score |
Candidates score |
|
1. |
12 |
|
|
2. |
12 |
|
|
3. |
14 |
|
|
4. |
14 |
|
|
5. |
13 |
|
|
6. |
15 |
|
|
|
80 |
|

QUESTIONS
-
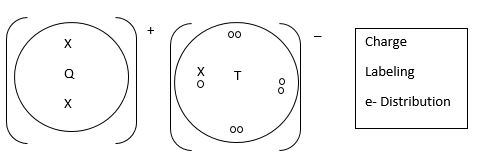
- The table below gives information about elements Q, R, S and T. The letters are not the actual symbols of the elements.
Element
Atomic number
Atomic radius (nm)
Ionic radius (nm)
Q
3
0.134
0.074
R
5
0.090
0.012
S
13
0.143
0.050
T
17
0.099
0.181
- In which period of the periodic table is element R? Give a reason. (2 mks)
- Explain why the atomic radius of R is smaller than that of Q. (2 mks)
- Explain why the ionic radius of T is larger than its atomic radius. (2 mks)
- Using dots (.) and crosses (x) to represent the outermost electrons, draw a diagram to show the bonding in the compound formed when Q reacts with T. (2 mks)
- The table below shows some properties of substances E, F and G. Study it and answer the questions that follow.
Substance
Melting point (ºc)
Solubility in water
Electrical conductivity
Solid state
Molten state
E
-39
Insoluble
Good
Good
F
1610
Insoluble
Poor
Poor
G
801
soluble
Poor
Poor
- Select a substance with:
Giant atomic structure ____________________________________________ (1mk)
Giant ionic structure ______________________________________________ (1mk) - Which substance is likely to have metallic bonding? Give a reason (2 mks)
- Select a substance with:
- The table below gives information about elements Q, R, S and T. The letters are not the actual symbols of the elements.
-
- Define the term molar heat of combustion. (1 mk)
- The following results were obtained when an experiment was carried out to determine the heat of combustion of ethanol. The experiment involved heating a known volume of water with a flame from ethanol burner. The burner was weighed initially and after the experiment.
Results
Volume of water in glass beaker = 200cm3
Mass of ethanol burner at the start = 86.66g
Mass of ethanol burner after experiment =86.20g
Initial temperature of water =24.0ºc
Density of water =1g/cm3
Specific heat capacity of water =4.2kj/kg/k
Calculate the:- Mass of ethanol burned (1 mk)
- Amount of heat produced in this experiment (2)
- Moles of ethanol burned ( C=12, H=1, 0=16) (2 mks)
- Molar heat of combustion of ethanol (2 mks)
- write a thermochemical equation for the combustion of ethanol (1 mk)
- Draw an energy level diagram for the combustion of ethanol. (3 mks)
-
- Name the following compounds
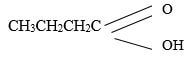 (1mk)
(1mk)- CH3COOCH2CH2CH3 (1mk)
- Name any two components of crude oil found in laboratory gas (2 mks)
- What is the difference between thermal cracking and catalytic cracking? (2 mks)
- Study the flow chart below:-
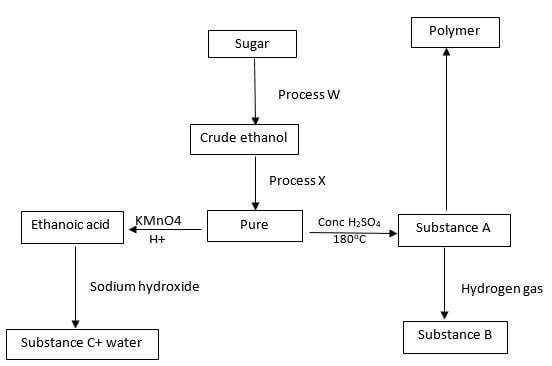
- Identify process W and X (1 mk)
- Name substances B and C (1 mk)
- Write the equation for the reaction leading to production of substance A (1 mk)
- Substance B was completely burned in air. Write the equation for the reaction (1 mk)
- State and explain the observation made when bromine is added to a gas jar full of substance B in the presence of sunlight (2 mk)
- The polymer D has a relative molecular mass of 112, 000. Calculate the number of monomers (C=12, H=1) (2 mks)
- Name the following compounds
-
- The diagram below shows a set up used in the electrolysis of molten copper (II) chloride using inert electrodes
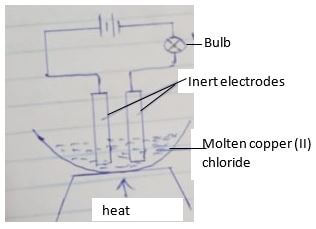
- What happens to the brightness of the bulb as electrolysis proceeds? Explain (1 mk)
- Label the cathode on the diagram above (1 mk)
- Write the half equation for the reaction at the anode (1 mk)
- Name a suitable pair of inert electrodes that can be used in this experiment
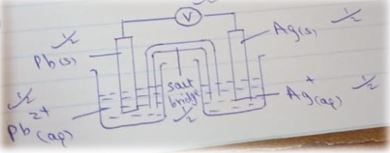
- Use the standard reduction electrode potentials given below to answer the questions that follows.
EØ (Volts)
Zn2+ (aq) + Ze- → Zn (s) – 0.76
Pb2+ (aq) + 2e– → Pb (s) -0.13
Cu2+ (aq) + Ze- → Cu(s) + 0.34
Ag+(aq) + e- → Ag(s) +0.80 -
- identify the strongest reducing agent. Give a reason (2 mks)
- Which two half cells would give the highest emf when combined? (1 mk)
- Calculate the end of the two half cells identified in (ii) above. (1 mk)
- Explain whether or not the following reaction can take place: (2 mks)
- Draw a well labelled diagram of the electrochemical cell obtained when the half cells of lead and silver are combined. (3 mks)
- The diagram below shows a set up used in the electrolysis of molten copper (II) chloride using inert electrodes
-
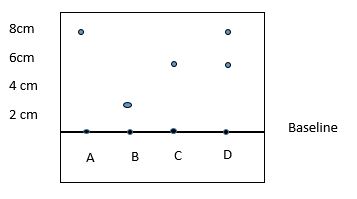
- The diagram below shows spot of pure A, B and C on chromatography paper. Spot D is that of a mixture.
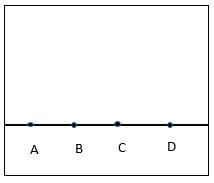
After development A,B and C were found to have moved 8cm, 3cm and 6cm respectively. D was separated into two sports which had moved 6cm and 8 cm.
On the diagram- Label the baseline (1mks)
- Show the position of all the spots after development (3mks)
- Identify the substances present in the mixture D (2mk)
- Describe how ammonium chloride can be separated from a solid mixture of ammonium chloride and anhydrous calcium chloride (3mk)
- The table below shows liquids that are miscible and those that are immiscible
Liquid L3 L4
L1 Miscible Miscible
L2 Miscible Immiscible
Use the information given to answer the questions that follow.- Name the method that can be used to separated L1 and L3 from the mixture of the two (1 mk)
- Describe how a mixture of L2 and L4 can be separated (3 mk)
- The diagram below shows spot of pure A, B and C on chromatography paper. Spot D is that of a mixture.
-
- The extraction of aluminum from its one takes place in two stages, purification stage and electrolysis stage.
- Name the ore from which aluminum is extracted. (1 mk)
- Name one impurity which is removed at the purification stage (1 mk)
- Name the substance added to the electrolyte to lower in order to its melting point. (1 mk)
- Write the equation for the reaction that takes place at:
Cathode (1 mk)
Anode (1 mk)
- Iron is extracted from its ore by the blast furnace process.
- Name the method used to concentrate the ore (1 mk)
- Name one ore from which iron is extracted (1 mk)
- One of the components of the waste gases from furnace is nitrogen (iv) oxide. Describe the adverse effects it has on the environment. (2 mks
- Give two uses of wrought iron (2 mks)
- During the extraction of zinc, the ore is first roasted in air before feeding it into a furnace.
- Name the main ore from which zinc is extracted. (1 mk)
- Write an equation for the reaction that takes place when the ore is roasted in air (1 mk)
- Give two uses of zinc metal (2 mks)
- The extraction of aluminum from its one takes place in two stages, purification stage and electrolysis stage.

MARKING SCHEME
-
-
- period 2. It has two energy levels.
- R has more protons, than Q hence stronger nuclear charge than Q which attracts electrons strongly towards the center.
- T form ions by gaining an electron, which is repelled by the already existing electrons thus increasing ionic radius.
-
giant atomic structure – F- E
It is a metal since it conducts electricity both in solid and molten state
-
-
- The enthalpy change when one mole of a substance is completely burnt in oxygen. (1 mk)
-
- 86.66-86.20
= 0.46g - DH=MCDT
= 200/1000 X 4.2 X 15
= 12.6kJ - RMM C2H5O4 = 46
Moles = 0.46/46 = 0.01
0.01 moles = 12.6kJ - 1 mole = 1x12.6/0.01
= -1260kJ/mol
- 86.66-86.20
- C2H5OH+302 2C02 +3H20 DH = -1260kJ/Mol
(l) (g) (g) (c) -
-
-
- butanoic acid
- Propyl ethanoate
- Propane, butane
- Thermal cracking occur in absence of catalyst at 700ºc
Catalytic occurs in presence of aluminum oxide catalyst at 400ºc 1450ºc -
- W- fermentation
X-fractional distillation - B – Ethane
C- Sodium ethanoate - CH3CH2OH → C2H4 + H2O
(g) (g) (l) - 2CH3CH3 +502 → 2 CO2 + 6H2O
(g) (g) (g) (l) - Orange bromine turns to colourless substance. A substitution reaction occurs between bromine and ethane.
- C2H4 = 28, no of monomers = 11200/28 = 4000
- W- fermentation
-
-
-
- Brightness recues, the concentration of the ions reduces since they are discharged at the electrons,.
- On the diagram
- 2cl- → Cl2 + Ze-
- Graphite/carbon (or platinum)
-
- Zn, it loses electrons most readily.
- Half cells of Zn and Ag.
- EØcell = 0.80 – (-0.076)
= +1.56V - EØ cell = - 0.76 –(0.34)
=M-1.10V
The reaction cannot occur since EØ is negative. -
-
-
-
- Substance A and B
- Heat the mixture. Ammonium chloride will sublime and deposit on the cooler part of the boiling tube. Calcium nitrate will remain at the bottom of the boiling tube.
-
- Fractional distillation
- Pour both liquids in separating funnel and allow it to settle, the denser liquid will settle down and less dense, liquid will form the top layer. Open the tap and run the liquid in the bottom layer leaving the liquid in the second layer.
-
-
-
- bauxite
- Iron (iii) oxide or silicon (iv) oxide
- Cryolite
- Cathode
Al3+ + 3e- → Al
(l) (l)
Anode
2O2- → O2 + 4e-
(l) (g)
-
- Froth floatation
- Iron pyrites
-
- Its poisonous causing respiratory diseases
- Causes acid rain which corrodes building etc.
- Making nails
- Making construction beams.
-
- Zinc blende
- 2ZnS + 302 → 2ZnO +2 SO2
(s) (g) (s) (g) -
- galvanizing iron sheets
- Manufacture of dry cells. etc.
-
Download Chemistry Paper 2 Questions and Answers - Nyeri Mocks 2021 Exams.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students