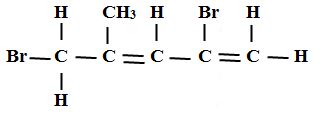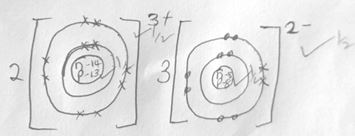Questions
Instructions to the candidates:
- Answer all the questions in the spaces provided.
- All working must be clearly shown where necessary.
- Mathematical tables and electronic calculators can be used.
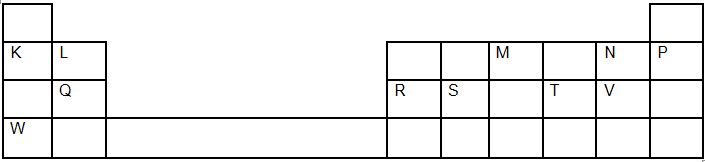
- The grid below shows a section of the periodic table, the letters are not the actual chemical symbol.
- Name the family into which element P belongs to ( 1mk)
- Which two elements forms the most soluble carbonates (2mks )
- With a reason, identify elements in period 3 with the largest atomic radius (2mks )
- Write the formula of the compound formed between Q and M (1mk )
- State two uses of element R and for each use , state property of element R that makes lts possible for the use
- Use ( 1mk)
- Property (1mk)
- Use (1mk)
- Property (1mk)
- Using dots and cross ,show bonding in the compound formed between R and oxygen (2mks )
- In terms of structure and bonding explain why the oxides of element Thas relatively low boiling points (2mks)
-
- name the following compounds (3mks)
- CH3CH2CH2COOH
-
- CH3CH2OOCCH2CH3
- Two types of detergents P and Q can be represented as
P: R -COONa- Identify each type of the detergent (2mks)
- Which of the two detergents is the best to use with hard water? Give a reason (2mks)
- State one advantage of detergent P (1mk)
- State one disadvantage of detergent Q (1mk)
- An hydrocarbon can be represented as follows
- Identify the hydrocarbon (1mk)
- Name two reagents that can reacted together to generate the hydrocarbon (2mks)
- name the following compounds (3mks)
-
- Name two apparatuses that can be used for determining mass in a laboratory (2mks)
- One of the flames produced by Bunsen burner is the luminous flame
- Explain why this flame is very bright (1mk )
- State two disadvantages of the luminous flame (2mks)
- Air is usually one of the substances that is considered as a mixture
- Identify the two most abundant component of air (2mks )
- Give two reasons why the air is considered as a mixture (2mks)
- One of the components of air is carbon (iv) oxide. Describe an experiment that can be used to prove the presence of carbon (iv) oxide in the air (2mks)
-
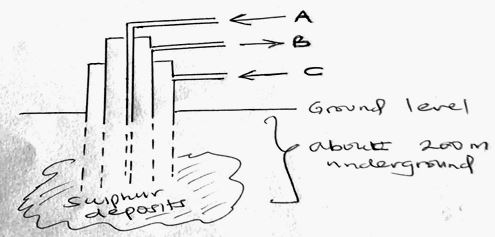
- The diagram below shows the process used to obtain Sulphur from underground deposits
- Name the above process used to obtain sulphur from the underground deposits (1mk)
- Name the substance passed through pipe
- (1mk)
- (1mk)
- State two properties of Sulphur that makes it possible to extract using the above process (2mks)
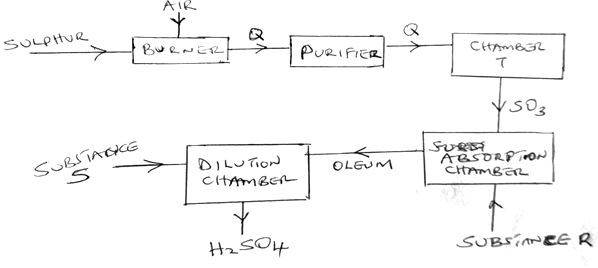
- The diagram below shows the contact process used in the manufacture of concentrated sulphuric(vi) acid
- Identify the following:
- Substance Q formed in the burner (1mk)
- Chamber T (1mk)
- Substance R (1mk)
- Substance S (1mk)
- Write the chemical equation occurring in the dilution chamber (1mk)
- Why is it necessary to pass substance Q though a purifier (1mk)
- State one use of sulphuric (VI) acid (1mk)
- Identify the following:
- The diagram below shows the process used to obtain Sulphur from underground deposits
-
- Calamine is one of the ores from which zinc can be extracted from
- Name any other ore from which zinc can be extracted from (1mk)
- The calamine is usually decomposed by heating to obtain substance M as shown below
ZnCO3 → M + CO2
Identify substance M (1mk) - Identify two methods that can be used to obtain zinc from substance M (2mks)
- During the extraction of zinc, name two gases likely to emitted into the air and that are likely to cause pollution (2mk)
- State one likely pollution effects of each of the gases you have mentioned in (a) above (2mks)
- State one possible use of zinc metal (1mk)
- Calamine is one of the ores from which zinc can be extracted from
-
- define the term electrolysis (1mk)
- State two functions of a salt bridge during electrolysis (2mks)
- The reduction potential of elements K, L, M, and P are as given below.
K+(aq) + e- ⇌ K(S), E = -1.46v
L2+ + 2e- ⇌ L(S) , E = +0.49V
M2+ ⇌ M(S), E = -2.69V
N+(aq) + e- ⇌ N(s), E = +0.52 V
P+(aq) ⇌ P(s), E= - 0.86V- Which letter represents the, strongest reducing agent? give a reason (2mks)
- Which two letters represent elements whose half cells would form an electrochemical cell with the largest e.m.f? (1mk)
- Calculate the e.m.f of the cell formed in (ii) above (2mks)
- During the electrolysis of a molten chloride of metal Q, a current of 0.25A was passed though the molten chloride for 2 hours and 10minutes. Given that 0.9grams of metal Q were deposited at the cathode.
- Calculate the quantity of electricity passed (1mk)
- Charge carried by the ions of metal Q given that R.A.M of metal Q is 84 (3mks)
-
- starting with magnesium oxide, describe how you can obtain a dry sample of magnesium
Carbonate (3mks) -
- Give one example of an acid salt ( 1mk)
- When sodium nitrate was heated a solid A and gas B were produced identify solid A and gas B (2mks)
- State two uses of gas B produced in (ii) above (2mks)
- State two factors that should be considered when choosing a fuel (2mks)
- starting with magnesium oxide, describe how you can obtain a dry sample of magnesium
Marking Scheme
-
- Noble gases
- K and W
- Q, it has lowest nuclear charge hence electrons in the energy level are least pulled towards the nucleus
- Q3M2 or Mg3N2
-
- Used for making sufuria /cooking pan
Property – good conductor of heat - Used for making overhead cables
Property – not easily corroded / good conductor of electricity (penalize electrical cables)
- Used for making sufuria /cooking pan
-
- It has molecular structure with weak van der waals forces of attraction between the molecules which require little energy to break.
-
-
- Butanoic acid
- 2,5 _ dibromo -4-methylpent-1,3-diene
- Enthyl propanoate
-
- P-soapy detergent
Q – soaplesss detergent - Q – does not form scum with hard water or it lathers easily
- It is biodegradable
- It is non-biodegradable hence pollutes the environment
- P-soapy detergent
-
- Ethene
- Ethanol and concentrated sulphuric (VI) acid
-
-
-
- Top pan balance
- Electronic balance
- Beam balance
-
- Due to incomplete combustion, it produces white hot carbon particles that emittes a lot of light
- It produces soot that makes apparatus dirty
- It does not produce much heat
-
- Nitrogen and oxygen
- It can be separated by physical means
Components of air are not chemically combined - Pass air through lime water (Ca(OH)2) the lime water forms white precipitate indicating presence of carbon(IV)oxide
-
-
-
- Frasch process
- A – hot compressed air
C – super heated water - It has low boiling point
It is insoluble in water
-
- Sulphur (IV)oxide
- Catalytic chamber
- Concentrated sulphuric (VI)acid
- Water
-
- H2S2O7(l) + H2O(l) + H2SO4(l)
- To remove impurities which may poison the catalyst
-
- Manufacture of fertilizer
- Manufacture of detergent
- Manufacture of dyes and paints
- Used in lead acid accumulators
(any one correct)
-
-
-
- Zinc blende (penalize zinc sulphide )
- ZnO
- Reduction using carbon or carbon (II) oxide
- It is converted to zinc sulphate and electrolyzed
-
- Sulphur {IV} oxides/SO2
- Carbon {IV} oxide /CO2
-
- Sulphur {IV} oxide leads to formation of acid rain
- Carbon oxide causes global warming
-
- Zinc is used to galvanise iron to prevent it form rusting
- To make brass an alloy of copper and zinc {any one correct}
-
-
- Electrolysis is the chemical decomposition of an electrolyte using electrical energy
-
- Complete the circuit by making contact between the two solutions
- Maintains balance of charges in electrolytes by providing ions to replace those that are used up or those that are formed
-
- M – it has the most negative E^θ value
- M and N
- Eφ =Eφ R-h-s
+0.52 – [-2.69]
=+3.21v - Q = 1 t
=0.25 x 130x60
=1950C - 1950C →deposits 0.9g
←84g
84 x 1950
0.9
=182,000C
1 F → 96,500C
← 182,000C
182000x1
96500
1.88 =2F
Charges is 2+
-
-
- Add magnesium oxide to HNO3/HCL/H2SO4 till in excess
- Filter to obtain the filtrate
- Add Na2CO3[any soluble carbonate] solution
- Filter to obtain insoluble magnesium carbonate
- Rinse and dry between filter papers
-
- NaHSO4 /KHSO4
- Solid A – NaNO2/Sodium nitrate
Gas B – O2/oxygen -
- Mixed with helium is used by mountain climbers and deep sea divers
- Air enriched with oxygen is used in hospitals by patients with breathing difficulties
-
- Availability
- Cost of fuel
- Heating value
{any two correct}
-
Download Chemistry Paper 2 Questions and Answers - Asumbi Girls High School KCSE Mock 2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students