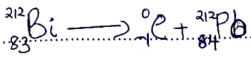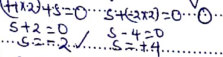Instructions to Candidates
- Answer ALL the questions in the spaces provided below each question.
- Mathematical tables and silent electronic calculators may be used.
- All working MUST be clearly shown where necessary.
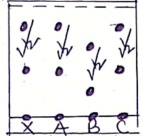
- Use the diagram below to answer the questions that follow.
- In which set – up will the iron nail rust? Explain (2 marks)
- State one advantage of rusting (1 mark)
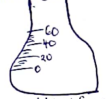
- During distillation in a laboratory the distillate can be collected either by a beaker or a conical flask.
- Define the term distillate. (1 mark)
- Explain why a conical flask is the most preferred apparatus for the collection of the distillate. (1 mark)
- Draw the diagram of a graduated conical flask. (1 mark)
- Describe how you would test for pH value of plant extract (3 marks)
- During investigation to identify illegal steroids used by three athletes. Urine samples were obtained and labeled as A, B and C. Illegal steroid was labeled as X, each urine sample had 2 components each and urine A and C contain the illegal steroid.
- Give the name of process used to identify illegal steroids in athletes. (1 mark)
- Draw a chromatogram, showing the information given above (2 marks)
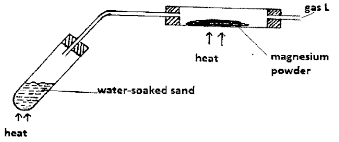
- The following set up was used to react steam with Magnesium Powder.
- Explain why the water-soaked sand was heated before heating the iron powder. (1 mark)
- Write an equation for the reaction that took place between steam and magnesium powder. (1 mark)
- State how gas L would be collected without using water. (1 mark)
-
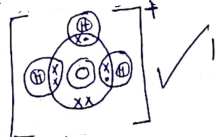
- Using dots(•) and crosses(x) ,show bonding in Hydroxonium ion, (H3O+) (H =1.0,O = 16.0) (1mark)
- The chloride of Aluminum was added to water and stirred. State and explain the observation made when two drops of methyl orange were added into the solution. (2 marks)
- The data below gives the electronic configuration of some selected atoms and ions
Atom/ion A2+ B C2− D2+ E F G+ H Electronic configuration 2 2.4 2.8 2.8.8 2.8 2.8.8 0 2.8.2 - Select an atom that is a noble gas (1 mark)
- What is the atomic number of: (1 mark)
- Select an element that belongs to group II and period 4 (1 mark)
- Use the table below to answer the questions that follow:
Element Atomic number A 11 B 13 C 14 D 17 E 19 - Write an equation for the reaction between element A and water. (1 mark)
- Compare the atomic radii of elements A and D. Explain. (2 marks)
- Starting with Copper metal describe how to prepare pure Copper (II) sulphate crystals. (3 marks)
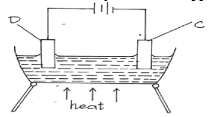
- The diagram below was used to electrolyze molten copper (II) chloride using graphite electrodes.
- Explain the role of heat on the above set up. (1 mark)
- Write an equation for the reaction taking place at electrode: (2 marks)
- C………………………………………………………………………………………………….
- D……………………………………………………………………………………………………..
- A state of equilibrium between dichromate (VI) and Chromate ions is established as shown below.
Cr2O72-(aq) + 2OH-(aq)CrO42-(aq) + H2O(l)
(Orange) (Yellow)- What is meant by a dynamic equilibrium? (1 mark)
- State and explain observation made when a few pellets of Potassium hydroxide are added to the equilibrium mixture. (2 marks)
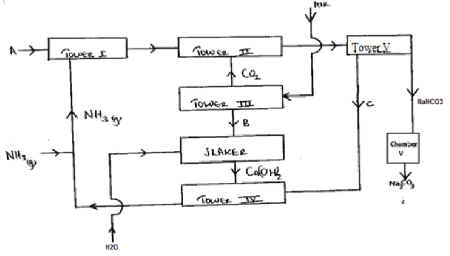
- The figure below shows some steps in the industrial preparation of sodium carbonate. Study it and answer the questions that follow.
- Identify substances labelled B and C (1 mark)
- Name the method that is used to separate sodium hydrogen carbonate and ammonium chloride. (1 mark)
- Name the most suitable substance used in Tower III to produce Carbon (IV) oxide. (1 mark)
- Calculate the relative formula mass of gas A given that the time taken for equal volumes of oxygen and gas A to diffuse through the same hole is 20 seconds and 24 seconds respectively (O=16.) (2 marks)
- Propane gas was bubbled through bromine liquid in a boiling tube.
- State condition required for the reaction to occur. (1 mark)
- Name the type of the reaction above. (1 mark)
- State one observation made during the reaction. (1 mark)
- Zinc metal and hydrochloric acid react according to the following equation.
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
2.0g of zinc were reacted with 100cm3 of 0.2M Hydrochloric acid,
Determine the reagent that was in excess (3 marks) -
- What is meant by the term allotropy? (1 mark)
- Which type of Sulphur is formed under the following conditions?
Conditions Type of Sulphur (i) Mixing sodium thiosulphate with dilute hydrochloric acid (½ mark) (ii) Saturating distilled with Hydrogen sulphide then exposed to air (½ mark) (iii) Pouring boiling Sulphur into cold water (½ mark) (iv) Below 96°C (½ mark)
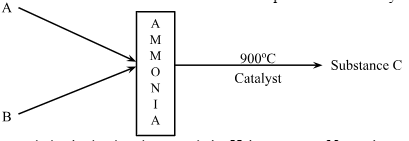
- The flow chart below shows the process that can be used to obtain substance C from ammonia gas when it is heated to about 900°C in air and in the presence of a catalyst.
- Ammonia is obtained on large scale by Haber process. Name the raw materials A and B. (1 mark)
- Name the substance C and the catalyst used. (1 mark)
- Write an equation to show the product formed when C is cooled in the presence of air. (1 mark)
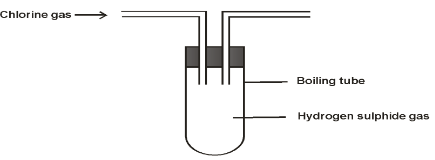
- In an experiment, Chlorine gas was passed into moist hydrogen Sulphide gas as shown below
- What observation was made in the boiling tube? (1 mark)
- Write an equation for the reaction which took place. (1 mark)
- What precaution should be taken in carrying out this experiment? Give a reason. (1mark)
- In an experiment to investigate the enthalpy of displacement of copper(II) ions by Zinc,50cm3 of copper(II) sulphate solution was placed in a plastic beaker. Excess zinc powder was then added into the beaker and allowed to react. After two minutes the highest change in temperature was noted to be 15°C
- Calculate the amount of heat given out during the reaction (C =4.2kJ/Kg/Density of solution = 1g/cm3) (1 mark)
- Given that the molar heat of displacement of reaction is −63kJ/mol,calculate the concentration of copper(II)sulphate solution in moles/litre.
(2 marks)
- The table below gives three experiments on the reaction of excess hydrochloric acid and 1.5g of zinc done under different conditions. In each the volume of gas was recorded at different time internals
On the axis below draw and label three curves that could be obtained from such results. (3 marks)Experiment Form of Zinc Hydrochloric acid solution I Powder 1.5M II Granules 1.0M III Powder 1.0M - An experiment was carried out where hydrogen chloride gas was bubbled through methylbenzene and water in separate beakers. The resulting solutions were tested with blue litmus papers and sodium carbonate
- Write the observations made in the following table (2 marks)
Solution of hydrogen Chloride gas in: Blue litmus paper Sodium carbonate Water Methylbenzene - Explain the observations in (i)above (1 mark)
- Write the observations made in the following table (2 marks)
- An element R has a relative atomic mass of 88. When a current of 0.5 A were passed through the fused chloride for 32 minutes and 10 seconds, 0.44g were deposited at the cathode. Determine the charge on the ion of R. (1 Faraday= 96500 C) (3 marks)
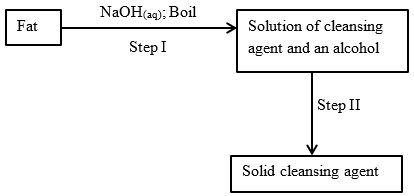
- The scheme below was used to prepare a cleansing agent. Study it and answer the questions that follow.
- What name is given to the type of cleansing agent prepared by the method shown in the scheme? (1 mark)
- Name one chemical substance added in step II (1 mark)
- What is the purpose of adding the chemical substance named in (ii) above? (1 mark)
- In the extraction of zinc, the zinc ore is crushed to a powder, mixed with oil and water and air blown through the mixture.
- What is the name given to this process? (1 mark)
- Explain how this process works. (1mark)
- Name the chief ore from which zinc is extracted. (1 mark)
- Radioactive polonium (Po) mass number 212 and atomic number 84 was detected in a sample of water. The water had an activity of 1000 counts per second.
- If the water is boiled explain whether the activity would be affected or not. (1 mark)
- Given that polonium resulted from Bismuth (Bi) following emission of a beta (β) particle; write a nuclear equation for the decay. (1 mark)
- State one medical application of radioactivity. (1 mark)
-
- State the Hess’s Law (1 mark)
- Calculate the molar enthalpy of formation of ethyne (C2H2) given the following. (2 marks)
C2H2 + 5/2O2(g) → 2CO2(g) + H2O(l) ΔH = −1300kJ/mol
C(S) + O2(g) → CO2(g) ΔH = −394kJ/mol
H2(g) + ½O2(g) → H2O(l) ΔH = −286kJ/mol
-
- Using the oxidation number, identify and explain oxidizing and reducing agent (2 marks)
2 H2S(g) + SO2(g) → 3S(s) + 2H2O(l) - Atomic number of Sulphur is 16. Write the electron configuration of S in SO32- (1 mark)
- Using the oxidation number, identify and explain oxidizing and reducing agent (2 marks)
MARKING SCHEME
-
- Iron is more reactive than coppper// Copper is less reactive than iron.
-
- Iron waste breaking down thus add nutrients to the soil
- Tightening of bolts and nuts of bridges.
-
- Condensed vapour collected after distillation.
- Has narrow mouth which ensures no spillage.
-
- Crush the plant extraxt using a pestle and mortar. Add propane/ acetone while stirring to obtain a solution. Decant and leave in the sun for propane/acetoone to evaporate. Add the universal indicator and match with the pH chart.
-
- Paper chromatography/chromatography.
-
-
- To generate steam that drives out ait initially in the combustion tube.
- Mg(s) + H2O(g) → MgO(s) + H2(g)
- Upward delivery/ Downward displacement of air. Accept - syringe
-
-
- Methyl orange changes to pink/red. Aluminium chloride hydrolyses in water to form hydrochloric acid/ acidic solution
-
-
- E
-
- A - 4
- C - 8
- D accept actual symbol
-
- 2A(s) + 2H2O(l) → 2AOH(aq) + H2(g)
or
2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) - D has a smaller size than A since D has more protons leading to more effective nuclear charge.
(A has larger atomic radius than D)
- 2A(s) + 2H2O(l) → 2AOH(aq) + H2(g)
- Heat the copper metal in an open crucible to obtain Copper(II) Oxide. Add excess CuO to warm dilute H2SO4. Filter to obtain Copper (II) Sulphate as a filtrate. Heat the solution to saturation and allow to cool to form CuSO4 crystals. Filter and dry between filter papers.
-
- To melt CuCl2 thereby setting up mobile ions
-
- C Cu2+(l) + 2e− → Cu(s)
- D 2Cl−(l) → Cl2(g) + 2e−
-
- State of balance in which the rate of forward reacion equals the rate of backward reaction,.
- Yellow colour intensity increases/ Orange colour fades. OH− concentration increases thus favours forward reaction/shifts equilibrium to the right
-
-
- B - Calcium oxide
- C - Ammonium chloride
- Filtration
- Calcium carbonate /limestone.
-
-

-
- Ultra Violet (UV) rays/sunlight
- Substitution.
- Brown Bromine liquid changes to colourless.
- Moles of Zn = 2.0 = 0.308 moles
65
Moles of HCl = 0.2 × 100
1000
= 0.02moles
0.02 moles of HCl requires
0.01 moles of Zinc; Zn is in excess. -
- Existence of an element in more than one form in the same physical state.
Conditions Type of Sulphur (i) Mixing sodium thiosulphate with dilute hydrochloric acid Colloiday (½ mark) (ii) Saturating distilled with Hydrogen sulphide then exposed to air Powdery (½ mark) (iii) Pouring boiling Sulphur into cold water Plastic (½ mark) (iv) Below 96°C Rhombic (½ mark)
- Existence of an element in more than one form in the same physical state.
-
- Nitrogen and Hydrogen
-
- Nitrogen(II)Oxide
- Platinum - Rhodium/Platinum
- 2NO(g) + O2(g) → 2NO2(g)
-
- Yellow deposits of Sulphur observed.
- H2S(g) + Cl2(g) → S(s) + 2HCl(aq)
- Done in fume cupboard/chamber. H2S is poisonous rej. in the open.
-
- ΔH = mcΔT
= 50 × 4.2 × 15 = 3.15kJ
1000 - 63KJ → 1 mole
3.15kJ → ??
3.15 × 1 = 0.05 moles
63
Molarity = 0.05 × 1000
50
= 1.0Moles/litre
- ΔH = mcΔT
-
-
-
Solution of hydrogen Chloride gas in: Blue litmus paper Sodium carbonate Water Blue litmus paper changes to red. Bubbles of a colourless gas Methylbenzene Blue litmus paper remains blue. No bubbles of a gas - HCl(g) ionizes in water to give H+ since water is a polar aolvent while in methylbenzene it doesn't ionize since Methylbenzene is a non-polar solvent
-
- Mass deposited = R.A.M × Q
charge × IF
0.44 = 88 × (0.5 × (32×60)+10
charge × 96,500
0.44 = 88 × 965
charge × 96,500
Charge = 2+/R2+
(Accept ALT) -
- Soapy
- Sodium Chloride
- Precipitate the soap
-
- Froth Floatation
- Oil and air cause the Zinc to float and impurities to sink.
- Zinc blended/Calamine
-
- Not affected, (Temp change doesn't affect nuclear reactions)
-
-
- Sterilizing surgical instruments
- Kill concerous tissues
-
- Energy change in converting reactants to products is the same regardless of the route by which the chemical change occur.
-
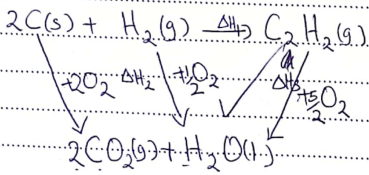
ΔH1 = ΔH2 − ΔH3
ΔH2 = (2x−394) + (11−286)
= − 1074
∴ ΔH1 = − 1074 − 1300
= − 1074 + 1300
= +226kJ/mol
-
-
2 H2S(g) + SO2(g) → 3S(s) + 2H2O(l)
↓ ↓ ↓
H2S is the reducing agent;oxidation number of S increases from −2 in H2S to O in S. SO2 is the oxidising agent; O.N of S decreases from +4 in SO2 to O in S. - S+(−2×3) = −2
S − 6 = −2
S = +4
16 − 4 =12
E.A is 2,8,2
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 1 Questions and Answers - Maranda High School Mock Exams 2023.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students