QUESTIONS
Instructions:
Answer all the questions in the spaces provided
- An element X exists in two isotopic forms i.e 10747X and n47X, If their relative abundance are in ratio of 3:1 respectively, calculate the atomic mass of the other isotope n47X given the relative atomic mass of X is 107.5. (3mks)
- Define the following terms. (3mks)
- Atomic number
- Mass number
- Ion
- Explain these chemical expressions that elements before reaction are electrically neutral. (2mk)
- The table below shows the atomic numbers of some elements represented by letters which are not their chemical symbols, Study it and answer questions that follow.
Element
J
K
L
M
N
P
Q
Atomic numbers
11
17
15
14
12
20
19
- Write down the electronic configuration of: (2mks)
- M
- P2+
-
- Write the formulae of the compound formed when K and M reacts (1mk)
- What type of bond is formed when K and M reacts. (1mk)
- How would reactivity of N and P with chlorine compare. Explain. (2mks)
- Select the most reactive metal and non-metal from the above list and give reason for your answer. (4mks)
- Metal
Reason - Non-metal
Reason
- Metal
- Place the elements in their immediate groups and periods.
Element
Group
Period
J
L
Q
- Write down the electronic configuration of: (2mks)
-
- Complete and balance the following chemical equations: (4mks)
Zn(s) + HCl(aq) →
Na2CO3 + H2SO4 →
Ca(OH)2 + HNO3 →
C2H6 + O2 → - Explain the following observations:
- Nitric acid is not used in preparation of Hydrogen gas by reacting metals and nitric acid. (2mks)
- Copper as metals does not react with dilute sulphuric acid. (2mks)
- Complete and balance the following chemical equations: (4mks)
- Using (.) and (x) notations show the bonding in MgCl2. (3mks)
- Suggest the type of bonds which is most likely to exist between the following compounds. (2mks)
- NaCl
- AlCl
- NH+4 ion
- Cl2(g)
MARKING SCHEME
Instructions:
Answer all the questions in the spaces provided
- An element X exists in two isotopic forms i.e 10747X and n47X, If their relative abundance are in ratio of 3:1 respectively, calculate the atomic mass of the other isotope n47X given the relative atomic mass of X is 107.5. (3mks)
R.A.M = isotopy × abundance
107.5 = (3/4 × 107) + 1/4 × n
107.5 = 80.25 + 0.25n
107.5 - 80.25 = 0.25n
27.25 = 0.25n
0.25 0.25
109 = n
n = 109 g/mol - Define the following terms. (3mks)
- Atomic number - no of protons in an element
- Mass number - sum of protons and neurons in an element
- Ion - charged particle
- Explain these chemical expressions that elements before reaction are electrically neutral. (2mk)
The elements have same number/magnitude of protons and electrons which negatively neutralize their effect - The table below shows the atomic numbers of some elements represented by letters which are not their chemical symbols, Study it and answer questions that follow.
Element
J
K
L
M
N
P
Q
Atomic numbers
11
17
15
14
12
20
19
- Write down the electronic configuration of: (2mks)
- M - 2.84
- P2+ - 2.88
-
- Write the formulae of the compound formed when K and M reacts (1mk)
K- + M4+ → MK4 - What type of bond is formed when K and M reacts. (1mk)
- Write the formulae of the compound formed when K and M reacts (1mk)
- How would reactivity of N and P with chlorine compare. Explain. (2mks)
- Select the most reactive metal and non-metal from the above list and give reason for your answer. (4mks)
- Metal O
Reason - Non-metal K - halogens are most reactive
Reason they have the highest electron affinity
for halogens affinity decreases down the group
- Metal O
- Place the elements in their immediate groups and periods.
Element
Group
Period
J
I 3 L
V 3 Q
I 4
- Write down the electronic configuration of: (2mks)
-
- Complete and balance the following chemical equations: (4mks)
Zn(s) + HCl(aq) → ZnCl2 (aq) + H2(s)
Na2CO3 + H2SO4 → NaSo4(aq) + CO2 + H2O(l)
Ca(OH)2 + HNO3 → Ca(No3)2 + 2H2O(l)
C2H6 + O2 → 4CO2(s) + 6H2O - Explain the following observations:
- Nitric acid is not used in preparation of Hydrogen gas by reacting metals and nitric acid. (2mks)
After reaction the hydrogen obtained is oxidised to water since nitric acid is a strong oxidising dilute agent - Copper as metals does not react with dilute sulphuric acid. (2mks)
Copper is unfer in reacting series than Hydrogen hence cant displace its form
- Nitric acid is not used in preparation of Hydrogen gas by reacting metals and nitric acid. (2mks)
- Complete and balance the following chemical equations: (4mks)
- Using (.) and (x) notations show the bonding in MgCl2. (3mks)
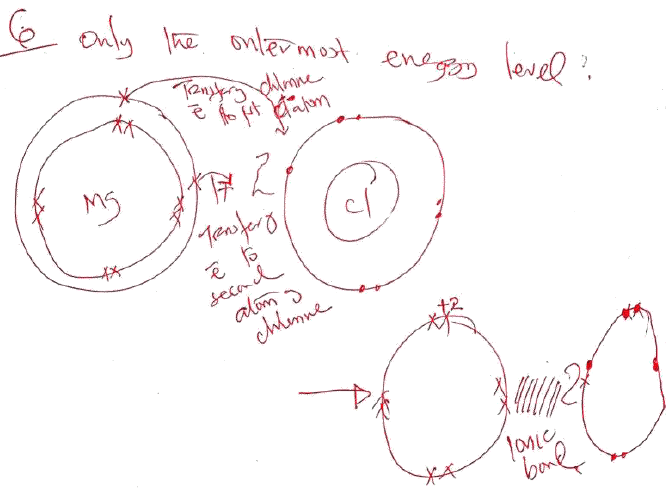
- Suggest the type of bonds which is most likely to exist between the following compounds. (2mks)
- NaCl - ionic bond
- AlCl3 - covalent bond
- NH+4 ion - co-ordinate bond
- Cl2(g) - covalent bond
Download Chemistry Questions and Answers - Form 2 Mid-term Exams Term 1 2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

