Chemistry Paper 2 Form 3 End Term 2 Exams 2021 with Marking Schemes
INSTRUCTIONS TO THE CANDIDATE
- Write your name, adm numbernumber and class in the space provided.
- Answer ALL the questions in the spaces provided.
- All working steps MUST be clearly shown, where necessary
- This paper consists of 8 pages
- Candidates should check the question paper to ascertain that all the pages have been clearly indicated and no question is missing.
- Hydrogen can be prepared by reacting zinc with dilute hydrochloric acid.
- Write an equation for the reaction. (1mk)
- Name an appropriate drying agent for hydrogen gas. (1mk)
- Explain why copper metal cannot be used to prepare hydrogen gas. (2mks)
- Hydrogen burns in oxygen to form an oxide.
- Write an equation for the reaction. (1mk)
- State two precautions that must be taken before the combustion begins and at the end of the combustion. (2mks)
- Give two uses of hydrogen gas. (2mks)
- When zinc is heated to redness in a current of steam, hydrogen gas is obtained. Write an equation for the reaction. (1mk)
- Element Q reacts with dilute acids but not with cold water. Element R does not react with dilute acids. Elements S displaces element P from its oxide. P reacts with cold water. Arrange the four elements in order of their reactivity, starting with the most reactive. (1mk)
-
- The number of protons, neutrons and electrons in atoms A to F are given in the table below the letters do not represent the actual symbol of the elements:-
Choose from the table the letters that represent:Atoms Protons Neutrons Electrons A 3 4 2 B 9 10 10 C 12 12 12 D 17 18 17 E 17 20 17 F 18 22 18 - An atom of a metal ....................................................... (1mk)
- A neutral atom of a non-metal ....................................................... (1mk)
- An atom of a noble gas ....................................................... (1mk)
- A pair of isotopes ................................................................... (1mk)
- A cation ............................................................................... (1mk)
- Anion …………………………………………... (1mk)
- The grid below shows a part of the periodic table. The letters do not represent the actual symbols. Use it to answer the questions that follow:-
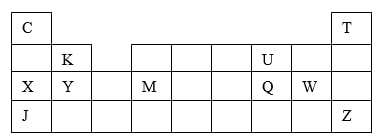
- How do the atomic radius of element
- X and M compare. Explain. (2mks)
- U and Q. Explain (2mks)
- Which letter represent the most reactive metal. Explain. (2mks)
-
- Using crosses (X) to represent electrons, draw the atomic structure of element Q (1mk)
- State the period and the group to which element Q belong (2mks)
Group ……………………………
Period ……………………………
-
- The ionic configuration of element G is 2.8 G forms an ion of the type G-1. Indicate on the grid, the position of element G. (1mk)
- To which chemical family does element G belong? (1mk)
- State one use of element U (1mk)
- What is the nature of the compound formed between K and U (1mk)
- How do the atomic radius of element
- The number of protons, neutrons and electrons in atoms A to F are given in the table below the letters do not represent the actual symbol of the elements:-
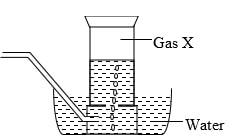
- Study the set-up below and answer the questions that follow:
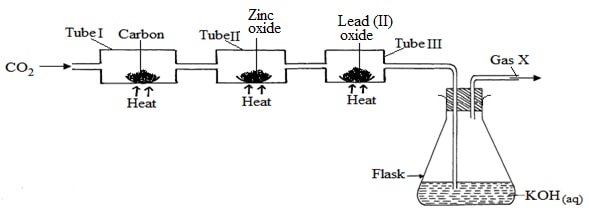
-
- Name Gas X ……………………………………………………………… (1mk)
- State the effect of releasing gas X to the environment (1mk)
- Write down equations for the reactions taking place in;
- Tube I (1mk)
- Tube II (1mk)
- Flask (1mk) ………………………………………………………………………………………………………
- State the observation made in tube III (1mk)
- Write down an equation for the reaction which could be used to generate Carbon (IV) Oxide for the above set up (1mk)
- Name the reagents used to generate gas X in the laboratory (1mk)
- Complete the diagram above to show how excess gas X can be collected (1mk)
-
- The flow chart below illustrates two industrial processes, Haber process and the Contact process:
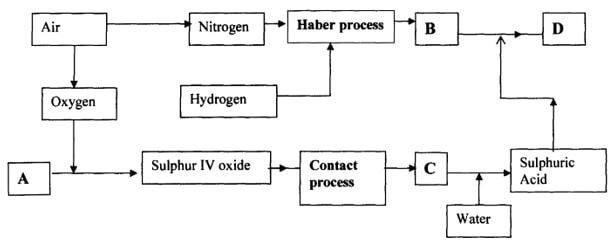
-
- Give the name of the process by which air is seperated into oxygen and nitrogen (1mk)
- Apart from oxygen and nitrogen gases produced from process (a)(i) name one other gas produced (1mk)
- Name the substances represented by the letters A, B, C and D (4mks)
- A …………………………………………..
- B …………………………………………..
- C ………………………………………….
- D ………………………………………….
- Name the catalysts used in:
- Haber Process …………………………………………… (1mk)
- Contact Process ……………………………………….. (1mk)
- Explain the role of the catalysts in both the Haber and the Contact processes (1mk)
- Write a chemical equation for the formation of compound B (1mk)
- Calculate the percentage by mass of the nitrogen present in compound D (2 mks)
- Give one major use of compound D (1mk)
-
-
- Two reagents that can be used to prepare chlorine gas are manganese (IV) oxide and concentrated hydrochloric acid.
- Write an equation for the reaction (1mk)
- Give the formula of another reagent that can be reacted with concentrated hydrochloric acid to produce chlorine gas (1mk)
- Describe how the chlorine gas could be dried and collected in the laboratory (2 mks)
- In an experiment, dry chlorine gas was reacted with aluminium as shown in the diagram below
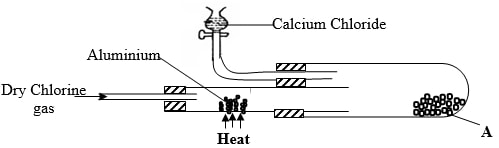
- Name substance A (1mk)
- Write an equation for the reaction that took place in the combustion tube (1mk)
- State the function of the calcium chloride in the set-up above (1mk)
- Two reagents that can be used to prepare chlorine gas are manganese (IV) oxide and concentrated hydrochloric acid.
-
- Sulphur occurs naturally in two different forms called allotropes;
- What are allotropes (1mk)
- The two allotropes of sulphur are stable at different temperatures, as shown in the equations below.
Give the name to the temperature 95.5ºC (1mk)
- Below is a flow diagram for the contact process for manufacture of Sulphuric acid(VI)
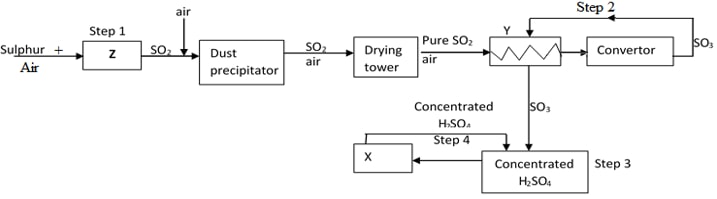
- Give the name of the chambers labelled (3mks)
Z …………………………………………
X ……………………………………………
Y …………………………………………… - State the three conditions in the converter (1mks)
- Explain why the gases are passed though:
- The dust precipitator and drying tower (1mk)
- The chamber labeled Y (1mk)
- Write the balanced equations for the reactions in :
Step 2 (1mk)
………………………………………………………………………………………………………
Step 3 (1mk)
………………………………………………………………………………………………………
Step 4 (1mk)
………………………………………………………………………………………………………
- Give the name of the chambers labelled (3mks)
- Sulphur occurs naturally in two different forms called allotropes;
- The following flow chart shows the industrial manufacture of Nitric (V) acid.
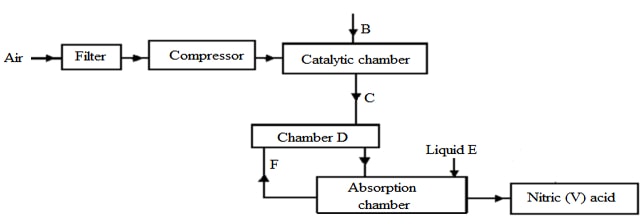
- Identify substance B, C, E and F. (4mks)
- B ………………………………………………….
- C ………………………………………………….
- E ………………………………………………….
- F ……………………………………………….....
- Describe what happens in the catalytic chamber. (2mks)
- State what takes place in chamber D. (1mk)
- 60 – 65% nitric (V) acid is produced in the absorption chamber. Describe how the acid can be concentrated. (2mks
- State why nitric (V) acid is stored in dark bottles. (1mk)
- Copper reacts with nitric (V) acid and not hydrochloric acid. Explain. (1mk)
- Identify substance B, C, E and F. (4mks)
Marking Scheme
-
- Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g) ✓1
- Concentrated sulphuric (IV) acid or anhydrous calcium chloride. ✓1
- Copper cannot displace hydrogen from its solution. ✓1
-
- 2H2(g) + O2(g) → 2H2O(l) ✓1
- Before: Pass hydrogen / through the tube before lighting ✓1 to drive off air.
End: There should be a continuous flow of hydrogen after / putting off the flame to avoid an explosion. ✓11
- – Filling balloons ✓1
- Manufacture of margarine.
- Manufacture of ammonia.
- Conversion of coal to synthetic petrol. - Zn(s) + H2O(g) → ZnO(s) + H2(g) ✓1
- S,✓½ P, ✓ ½ Q, ✓½ R, ✓½
- It adds to unsaturated oils and hardens them. ✓1
-
-
- C
- D or E
- F
- D or E
- A
- D
-
-
- Atomic radius of M is smaller than that of X. The effective nuclear charger in M is greater than in X hence outer electrons strongly pulled to the centre reducing the radius.
- Radius of Q is greater than that of U because Q has more energy levels than U
- J because it loses energy more easily due to the bigger atomic radius of J
-
- Period – 3 Group – VI
-
- On the grid (period 2 Group 7)
- Halogen
- – Used in hospitals with patients with breathing difficulties
- Used by mountain climbers and deep sea divers - Basic oxide
-
-
-
-
- Carbon (II) Oxide or CO – (reject Carbon monoxide)
- Combines with haemoglobin to form carboxyhaemoglobin which prevents carrying of oxygen
-
- CO(g) + C(s) →2CO(g)
- ZnO(s) + CO(g) → Zn(s) + CO2(g)
- KOH(aq) + CO2(g) → K2CO3(aq) + H2O(l)
- Orange/yellow Lead (II) Oxides turns grey
- CaCO3(s) + 2HCl(aq) → CaCl2(aq) + CO2(g) + H2O(l)
- Methanoic acid and concentrated sulphuric acid
-
-
-
-
- Fractional distillation of liquid air
- Argon
- A - Sulphur
B - Ammonia gas
C - Oleum
D - Amonium sulphate -
- Finely divided iron
- Vanadium (v) Oxide
- Speeds up the rate of reaction by lowering the activation energy
- 2NH3(g) + H2SO4(aq) → (NH4)2SO4(aq)
- R.M.M of (NH4) = 132
Mass of N = 28
% N = 28/132 x 100 = 21.212 % - Used as a fertilizer
-
-
-
- MnO2 (s) + 4HCl(l) → MnCl2(aq) + 2H2O + Cl2(g)
Penalize ½mk if state symbols are not correct - KMnO4 or PbO2
- The Chloride gas can be dried by passing it through a wash-bottle of concentrated sulphuric acid and is then collected by downward delivery.
- MnO2 (s) + 4HCl(l) → MnCl2(aq) + 2H2O + Cl2(g)
-
- A- Aluminium (III) Chloride
- 2Al(s) + 3Cl2(g) → 2AlCl3(s)
Penalize ½mk for wrong state symbols
-
-
-
- They are different physical/ structural forms of an element in the same physical state.
- Transition temperature
-
- X - Diluter
Y- Heat exchanger
Z- Roaster/ Burner - Catalyst- Vanadium (v) Oxide, V2O5
Temperature – 450C
Pressure – 1 atmosphere -
- - They are purified not to poison the catalyst
- - The reaction in the convertor/ production of sulphur (vi) Oxide is exothermic/heat is produced. Chamber Y is used to ensure temperature does not rise above 450oC
- Step 2: 2S02(g) +O2(g) → 2S03(g) ✓ 1 mark
Step 3: 503(g) + H2SO4(l) → H2S2O7(l) ✓ 1 mark
Step 4: H2S2O7(l) + H2O(l) → 2H2SO4(l) ✓ 1 mark
- X - Diluter
-
-
- B – ammonia gas ✓1
C - nitrogen (II) oxide (NO) ✓1
E – water ✓1
F – unreacted gases ✓1 - The mixture of ammonia and air is passed through heated/ catalyst where ammonia (II) is oxidized to nitrogen (IV) oxide. ✓1
- Gases are cooled and air passed through heated/ catalyst where ammonia is further oxidized to nitrogen(IV) oxide.✓1
- Fractional distillation, ✓
Water with a lower boiling point ✓ than nitric (V) acid, distills left leaving the concentrates acid. - HNO3 decomposes when exposed to light
- HNO3 is an oxidizing agent hence able to oxidizes copper to Cu2+
HCl on the other hand doesn’t react with copper because copper is less reactive and can not displace hydrogen from its acid.
- B – ammonia gas ✓1
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Form 3 Questions and Answers - End Term 2 Exams 2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

